SOLUTIONS FOR ISOLATED MYOCYTES
Cardiovascular disease is the leading cause of death in the industrialized world. The development of heart disease and, in turn, heart failure is most often a consequence of dysfunction within individual heart muscle cells, or cardiomyocytes. Individual cardiomyocytes represent the smallest fully functional model system of heart muscle that can be examined for ion regulation, force production, relaxation function, cell signaling, and gene expression.
Biochemical, molecular and hemodynamic changes can fundamentally alter the functionality of the cardiomyocyte, and hence understanding the regulatory factors in cardiomyocyte function is essential in the discovery and development of therapeutic interventions. Studies utilizing isolated cardiomyocytes have yielded important information regarding cardiomyopathies including, but not limited to, myocardial infarction, atrial fibrillation and sudden adult death syndrome.
IonOptix Systems for the study of isolated myocytes include…

MULTICELL SYSTEM
Our MultiCell System offers fully-automated identification and mechanical phenotype quantification from hundreds of myocytes per hour without sacrificing any of the detailed and thorough analysis of our standard systems. Featuring a fast x-y-z position-programmable scanning microscope coupled with a novel image analysis method quantifying position, size, and orientation, MultiCell brings dynamic characterization of contraction-relaxation function into the 21st century with greater statistical power, better acquisition and analysis, and a more user-friendly design. LEARN MORE
CALCIUM AND CONTRACTILITY SYSTEM
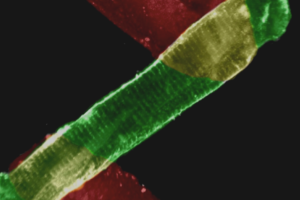
Striated muscle, like cardiac tissue, is characterized by the appearance of bands within the individual myocytes known as sarcomeres. The rates of sarcomere shortening and lengthening, as well as the magnitude of shortening, provide important indicators of contraction/relaxation dynamics. In addition, calcium is one of the most important regulatory factors in cardiac myocyte function, and is the link between the electrical signals which ripple through the heart and myocyte contraction. Changes in calcium release and handling in individual myocytes can dramatically affect the downstream mechanical activity of the myocyte. Our Contractility System and ever popular combination Calcium and Contractility System are cornerstones for reliable and repeatable quantification of cardiomyocyte contractile function. LEARN MORE

MYOSTRETCHER
Small changes in sarcomere length can produce large changes in tension development, known as length-dependent activation. Changes in sarcomere length are instrumental to regulation of contractile force, commonly known as the Frank-Starling Law. When a cardiomyocyte becomes stretched, for example with increased ventricular preload, the sarcomeres are also stretched resulting in an increase in the force of contraction. To improve the physiologic fidelity of our systems, we’ve developed additional tools to facilitate the attachment and mechanical loading (stretching) of intact isolated myocytes. The MyoStretcher combines motorized micromanipulators and a suite of components designed to simplify myocyte attachment, including a specially designed rotatable chamber. LEARN MORE