Go back to RESOURCES
Interpolated Numerator Method
Introduction
For dual excitation measurements with Fura-2, we offer two methods of collecting data, interleaved sampling and the interpolated numerator (or denominator) method. Interleaved is straightforward to explain: each wavelength is sampled in turn, and the maximum sampling rate is determined by how quickly the light source can switch. The interpolated numerator method works differently, which will be explained below. In addition we will provide some practical notes on doing experiments with the interpolated numerator method.
Isosbestic point
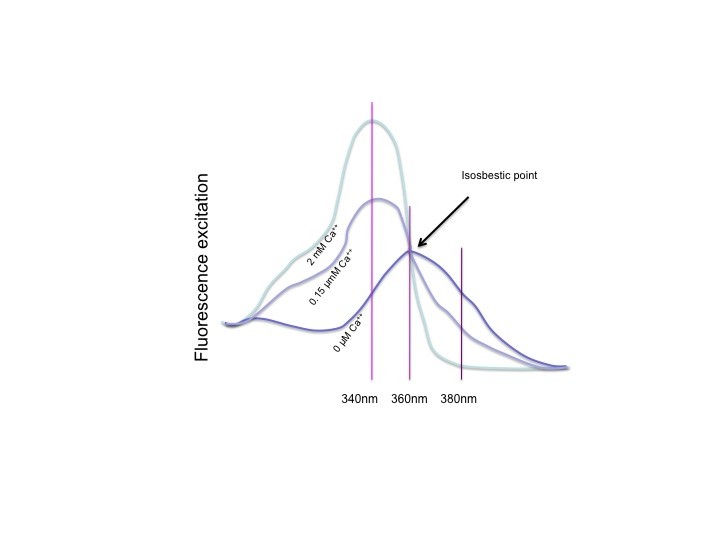
Fura-2 has an isosbestic point. The isosbestic point is the wavelength at which Fura-2free has exactly the same absorbance as Fura-2.Ca++. What it means is that the combined fluorescence of Fura-2free and Fura-2.Ca++ is the same at all calcium concentrations. Fluorescence at this excitation wavelength will only vary with the amount of Fura-2 that is excited. It will only change with the loss of Fura due to bleaching or leakage over the course of an experiment. These are relatively slow changes, and the interpolated numerator method takes advantage of that.
Interpolated numerator method
Because the changes in fluorescence at the isosbestic point only change slowly, it does not need to be sampled continuously. The sampling method we apply is illustrated in the middle figure. Data collection is started with measuring the isosbestic wavelength, 360nm, for a short period like 100 miliseconds. The 100 datapoints collected are averaged and the averaged value is stored. The light source then switches to 380nm, measuring the Fura-2Ca-free. This is done at up to the maximum frequency of the sensor, in the case of the PMT at a 1000Hz. At the end of the sampling epoch the light source briefly switches to the isosbestic point again, and collects another average value for the end of the epoch. All the data points for the numerator are now interpolated between the beginning and end value. The ratio is then calculated for each interpolated point and the corresponding measurement at 380 nm.
Advantages of the interpolated numerator method
Because the wavelength only needs to be changed twice in one run, from 360nm to 380nm at the beginning, and back to 360 at the end, high sampling speeds can be achieved (1000Hz) in spite of having relatively low speed wavelength switching. It thus allows the use of an economically price filterwheel. An extra advantage is that the ratio data will have relatively little noise, because the interpolated numerator is noise free where with interleaved sampling a noisy signal is divided by another noisy signal.
Reduced dynamic range
The drawback of the interpolated numerator method is a loss in dynamic range of the system. When sampling interleaved with 340nm and 380nm, an increase in calcium leads to an elevation at 340nm and a simultaneous drop at 380nm, resulting in a big change in the ratio. When the isosbestic point is used, the signal at 360 does not move, thereby also limiting the movement of the ratio.
Potential problems and remedies
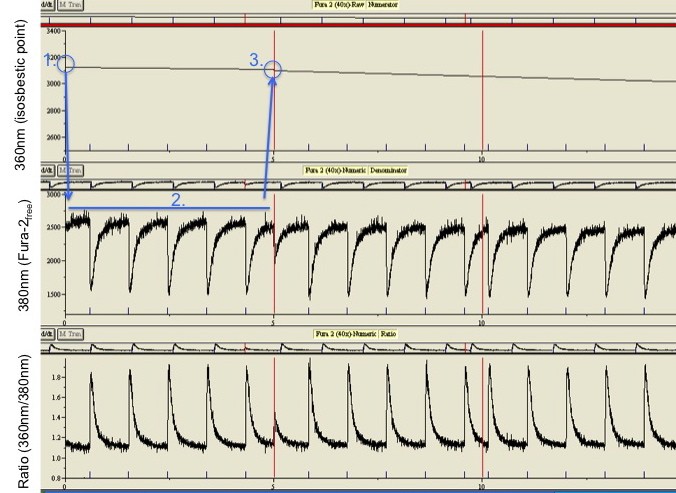
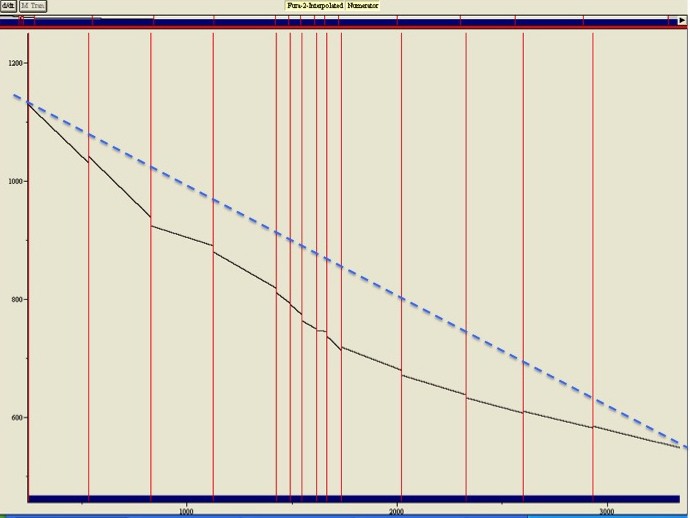
Bleaching is not linear. The figure on the right shows the fluorescence at the isosbestic point measured over 50 minutes. Over this time the fluorescence drops some 50% due to bleaching and expulsion of Fura-2 by the cell. Because the denominator will drop the same percentage, the ratio will be stable. This is exactly why ratiometric measurements are so powerful. However, as you can see with respect to the dotted line drawn across the figure, bleaching is not linear. If the isosbestic point had only been sampled at the beginning and end of the 50 minutes, the fluorescence in the middle of the experiment would have been grossly overestimated, resulting in an overestimation of calcium. For interpolation to be accurate, the interpolation point needs to be sampled regularly, in practice probably every 20-40 seconds.
The entire preparation must be in the field of view. The validity of interpolating data points is based on the assumption that the change in Fura-2 is predictable during an experimental run. Bleaching and transport out of the cell are predictable over a short period. But when the area that is sampled changes during the experiment, for example when not the entire preparation is in view and it starts to contract, the relation between numerator and denominator is lost and the ratios become nonsense. The reason the method works so well with isolated myocytes is because they are isolated, so they can be viewed in their entirety. The method would not work with a shortening muscle strip that is only partially in the field of view. Always make sure the entire cell is within the aperture of the diaphraigm, and don’t change the focus during a sampling run!